Dassault Systèmes Collaboration Yields Breakthrough Guide for Using Virtual Twins in Clinical Trials
Listen to this press release
- Findings were published from Dassault Systèmes’ five-year collaboration with the FDA in response to needs for faster, safer medical device evaluation
- The peer-reviewed report serves as a guide for establishing credibility in medical device in silico clinical trials
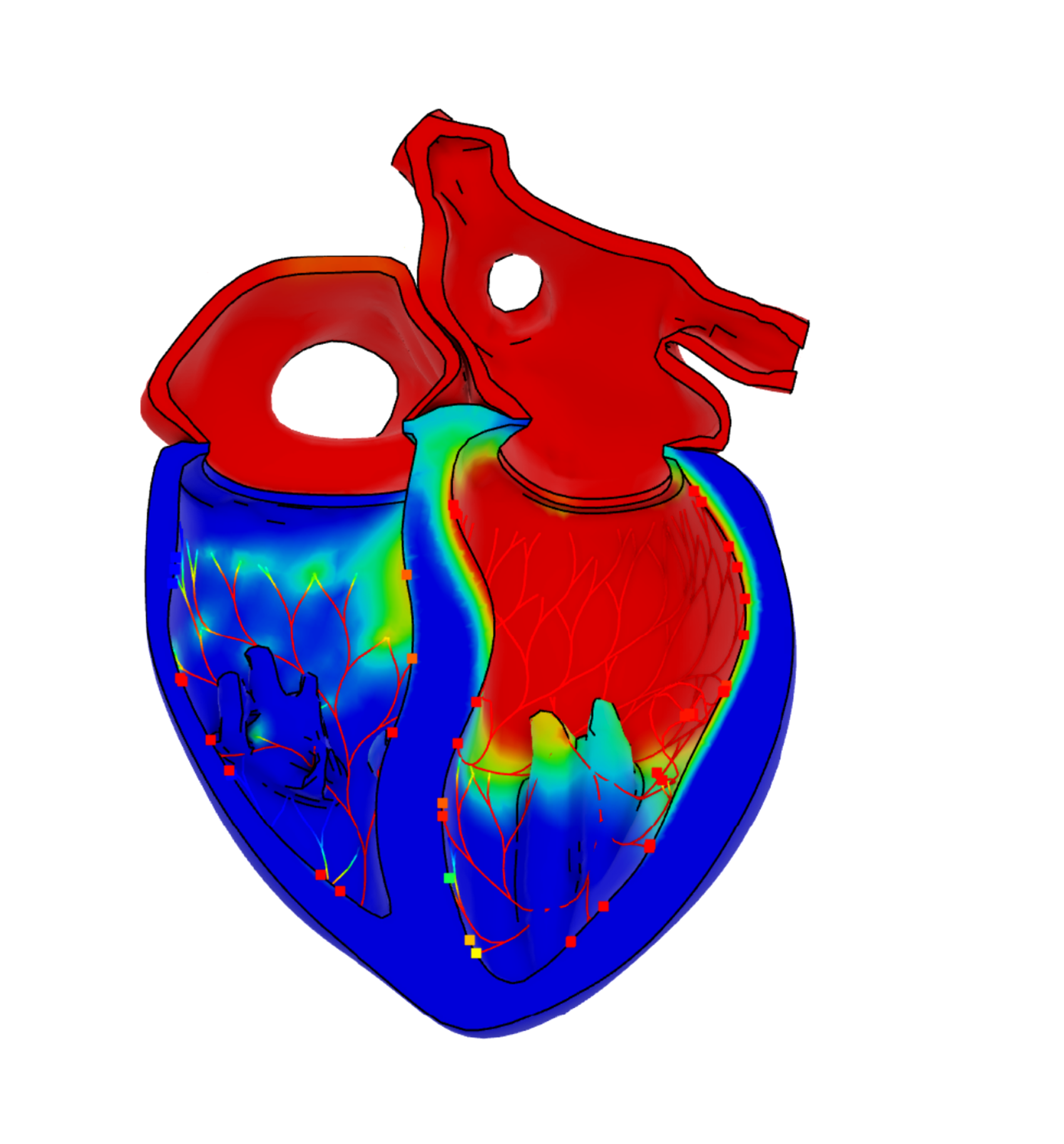
- Virtual twins that accurately simulate specific aspects of patient populations allow for the refinement, reduction and replacement of human and animal testing in clinical trials
VELIZY-VILLACOUBLAY, France — October 30, 2024 — Dassault Systèmes (Euronext Paris: FR0014003TT8, DSY.PA) today announced the availability of the world’s first guide for the medical device industry that outlines how to use virtual twins to accelerate clinical trials. This guide was published following the successful completion of a five-year collaboration with the U.S. Food and Drug Administration. The in silico clinical trial “ENRICHMENT Playbook” marks a significant advancement in the integration of virtual twins into the regulatory process in response to needs for improved patient safety, regulatory compliance, and pace of innovation.
The peer-reviewed, open-access 44-page publication serves as a comprehensive guide for establishing credibility in medical device in silico clinical trials by providing a hierarchical framework and strategic approach to overcome challenges. The project involved the creation of virtual twins that simulate patient populations with unprecedented accuracy – an innovative approach that allows for the refinement, reduction and replacement of human and animal testing. Industry can access knowledge on running trials, building patient populations, and interpreting and measuring results to better understand the effects of a device before it reaches a patient.
“As the healthcare industry continues to embrace digital transformation, the ENRICHMENT Playbook represents a pivotal moment in the evolution of clinical trial methodologies,” said Claire Biot, Vice President, Life Sciences and Healthcare Industry, Dassault Systèmes. “The strong participation and input from diverse experts was crucial in ensuring that the playbook meets the highest scientific and regulatory standards and is highly practical for immediate application in the field. We remain committed to driving collaborative innovation in this sector to improve the patient experience.”
The ENRICHMENT Playbook outlines the credibility assessment process based on recommendations from the recently issued FDA final guidance Assessing the Credibility of Computational Modeling and Simulation in Medical Device Submissions, and appears in the CDRH/OSEL Regulatory Science Tools Catalog. It is the result of extensive collaboration among leaders from the FDA’s Office of Science and Engineering Laboratories, clinical practice, academia, the medical device industry and regulatory science. The playbook’s authors are discussing outcomes and opportunities today at Dassault Systèmes’ International Virtual Human Twin Experience Symposium in Paris.
About Dassault Systèmes
Dassault Systèmes is a catalyst for human progress. Since 1981, the company has pioneered virtual worlds to improve real life for consumers, patients and citizens. With Dassault Systèmes’ 3DEXPERIENCE platform, 370,000 customers of all sizes, in all industries, can collaborate, imagine and create sustainable innovations that drive meaningful impact. For more information, visit: www.3ds.com
